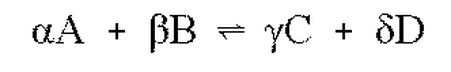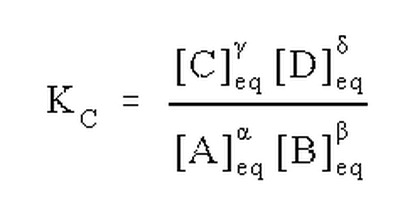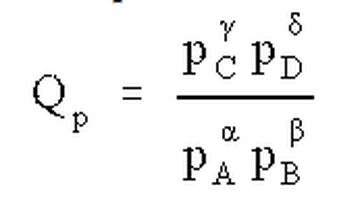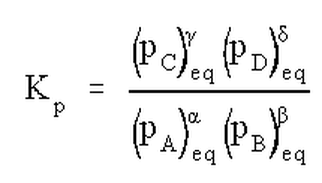The Reaction Quotient, Q
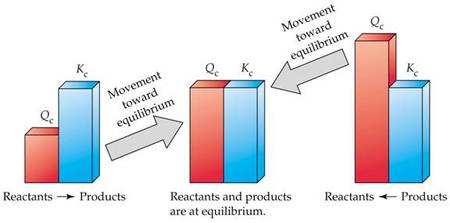
The ratio of the concentrations (or partial pressures) of products raised to their stoichiometric coefficients to the concentrations of reactants raised to their stoichiometric coefficients at any point in the reaction is called the reaction quotient, Q. Like K, Q can be expressed in terms of concentrations (Qc) or partial pressures (Qp). At equilibrium Q is equal to K; therefore, the direction in which a reaction will proceed can be determined by comparing Q to K. If Q < K, the reaction moves in the direction of the products; if Q > K, the reaction moves in the reverse direction.
The ratio of the concentrations (or partial pressures) of products raised to their stoichiometric coefficients to the concentrations of reactants raised to their stoichiometric coefficients at any point in the reaction is called the reaction quotient, Q. Like K, Q can be expressed in terms of concentrations (Qc) or partial pressures (Qp). At equilibrium Q is equal to K; therefore, the direction in which a reaction will proceed can be determined by comparing Q to K. If Q < K, the reaction moves in the direction of the products; if Q > K, the reaction moves in the reverse direction.
|
In general, the equilibrium constant is equal to the "proper quotient of equilibrium concentrations", but it does that mean? For every reaction, the equilibrium expression is is different. (The equilibrium expression is the equation that relates the equilibrium constant to some function of the reactant and product concentrations.) Suppose we right a general (but fictitious) reaction between two products and two reactants, with some set of stoichiometric coefficients which we will leave as greek letters
|
Note that the equilibrium constant may be quoted in terms of molarity, in which case it is called KC, or in terms of partial pressures, in which case it is called Kp. If we have a mixed set of standard states, or if we simply don't state what types of concentrations are used, we can call the equilibrium constant Keq or simply K.The choice of concentration units (or standard state) and all of its ramifications will be discussed in greater depth later. For now, we note how the equilibrium expression results from the balanced chemical reaction directly from the stoichiometric coefficients.